PRODUCTION AND FORMULATION OF BIOCONTROL AGENTS

BIOPESTICIDE PRODUCTION
Biopesticide active ingredients are obtained in microbial cultures in semi-industrial scale bioreactors. It consists of the optimization of process parameters to obtain cells, spores or fermentation metabolites. Medium composition, incubation time and growth conditions such as pH, temperature and aeration are optimized looking at the best characteristics required. Process development studies are carried out for subsequent production on an industrial scale.

BIOPESTICIDE FORMULATION
Optimized formulations are developed to ensure the activity of the biopesticide under conditions of commercial application, and to guarantee the stability during transport and throughout its shelf-life. Prototypes of liquid and dry formulations are improved to increase their stress tolerance. Dried formulations are obtained by dehydration using protectant agents allowing long-term storage and high cell viability.
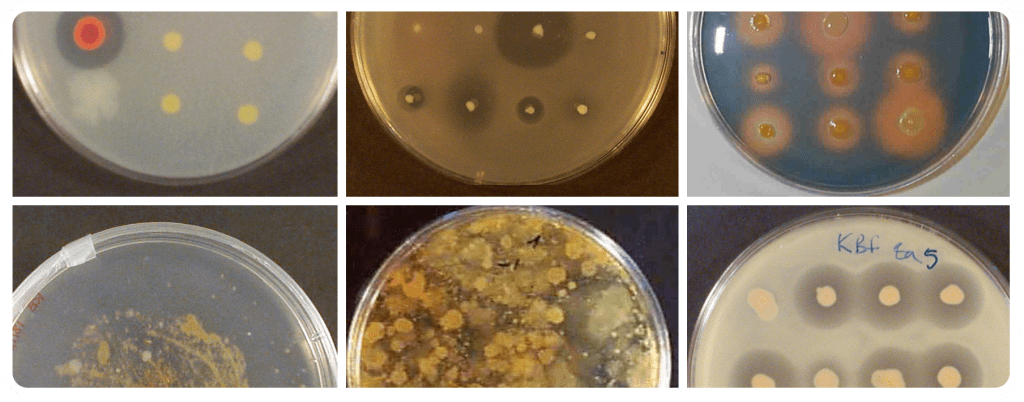
PRODUCT QUALITY CONTROL AND ACTIVE INGREDIENT MONITORING
At this stage, the quality of the obtained biopesticides is evaluated. Several physicochemical and/or microbial parameters of the biopesticide are determined. The shelf-life of the product is established considering microbial parameters that include the quantification of the active ingredient and the determination of antimicrobial activity against target pathogens. In the case of microorganisms, methods for quantifying cells by PCR (qPCR or v-qPCR) are developed, using strain-specific molecular markers to monitor the active ingredient over time.
